Abstract
Introduction: Mutations in isocitrate dehydrogenase 1 (IDH1) and IDH2 have been reported in clonal hematopoiesis and are recurrent, early events in the development of acute myeloid leukemia (AML). A subsequent stepwise acquisition of additional genetic aberrations leads to the development of frank leukemia. At the time of AML diagnosis, rare pre-leukemic hematopoietic stem cells (pHSC) can be detected containing some, but not all, of the mutations in the corresponding leukemia, although these pHSCs are phenotypically indistinguishable from residual wildtype hematopoietic stem cells (HSC). The frequency of pHSCs at the time of diagnosis is known to correlate to overall survival in AML patients (Corces et al, Nat Genet 2016), indicating the potential relevance of therapeutically targeting these cells to prevent relapse.
Methods: To model these rare pre-leukemic cells, we introduced IDH1 R132H and IDH2 R140Q mutants or their wildtype counterparts into human CD34+ hematopoietic stem and progenitor cells (HSPC) using CRISPR/Cas9 and AAV6-mediated homology directed repair (HDR). Successfully edited cells were then sorted by FACS based on GFP expression for subsequent analyses including 2HG quantification, colony formation, differentiation capacity, RNA sequencing, and metabolic assays. Additionally, primary pHSCs from diagnostic bone marrow aspirates from AML patients were also isolated by FACS and subjected to treatment, colony formation assays, and genotyping by ddPCR.
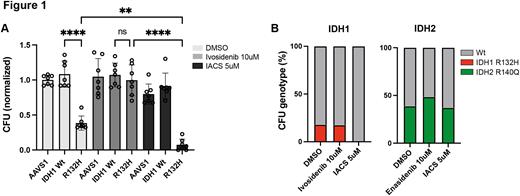
Results: Human CD34+ HSPCs engineered to express either IDH1 R132H or IDH2 R140Q produced 2HG and showed a striking reduction in colony formation and differentiation capacity, recapitulating the cytopenic phenotype observed in IDH-mutated clonal cytopenia of undetermined significance (CCUS). RNA-seq of these cells revealed large transcriptomic changes shared between IDH1 R132H and IDH2 R140Q mutations, but gene set enrichment analyses revealed the specific and significant downregulation of oxidative phosphorylation-related genes in R132H-mutated cells. These R132H cells were further confirmed to have lower basal metabolic activity coupled to a higher mitochondrial dependency as determined by SCENITH (Argüello et al, Cell Metab 2021). These metabolic alterations suggested that it might be possible to selectively therapeutically target R132H mutated cells. To evaluate this, we treated HSPCs engineered to express GFP, IDH1 wildtype, or IDH1 R132H, with the known mutant-specific inhibitor ivosidenib, the OXPHOS-inhibitor IACS-010759, or DMSO as a control. Strikingly, ivosidenib restored the defect in colony formation conferred by R132H and increased the total number of colonies whereas IACS-010759 effectively inhibited R132H colony formation (Figure 1A).
To further investigate this vulnerability, we sorted pHSCs from diagnostic bone marrow samples from primary IDH1 and IDH2-mutated AML samples according to the phenotype CD45dimCD3-CD19-CD20-CD34+CD38-CD99-TIM3-. These cells were subjected to treatment with DMSO, ivosidenib, enasidenib, or IACS-010759 and subsequently plated for colony formation. Because the sorted cell population contained a mixture of mutant pHSCs and residual wildtype HSCs, all colonies were picked and genotyped for IDH mutations using ddPCR, revealing that only IDH1 wildtype colonies grew out after IACS-010759 treatment whereas all other conditions resulted in a mixture of wildtype and mutant colonies (Figure 1B).
Conclusions: The OXPHOS-inhibitor IACS-010759 can eradicate IDH1-mutated pHSCs whereas ivosidenib cannot. This effect is specific for IDH1 mutations and the same effect is not seen in IDH2-mutated pHSCs. These results have potential clinical implications when considering therapeutic options in AML as well as IDH1-mutated pre-leukemic conditions such as CCUS.
Disclosures
Koehnke:TenSixteen Bio: Consultancy. Majeti:CircBio: Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees, Research Funding; Kodikaz Therapeutic Solutions: Membership on an entity's Board of Directors or advisory committees; Syros Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; TenSixteen Bio: Membership on an entity's Board of Directors or advisory committees; Gilead Sciences: Patents & Royalties: CD47 cancer immunotherapy, Research Funding; Pheast Therapeutics: Current equity holder in private company; MyeloGene: Current equity holder in private company; RNAC Therapeutics: Current equity holder in private company.
Author notes
Asterisk with author names denotes non-ASH members.